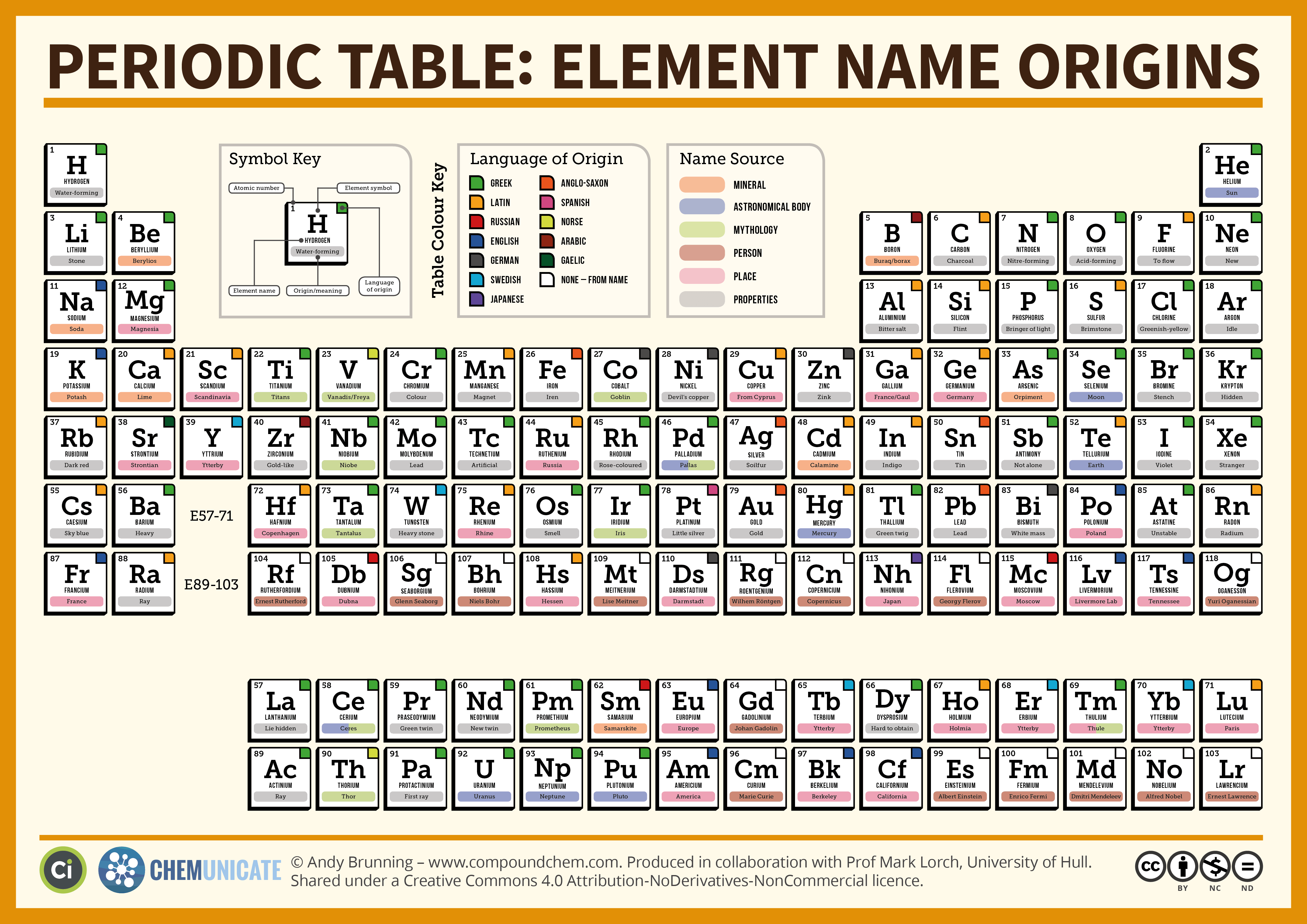 
A very early and detailed adoption of Moseley’s proposals for the employment of atomic numbers is to be found in the text of A Smith (1919), Intermediate Text Book of Chemistry, Century Co., New York. This eventually gave rise to the long form of the Table, and also to the many variants which are in circulation today. Atomic weights were regarded as a basis for systematization until after 1913, when the significance of atomic number and hence basic elemental electronic structure was described by Moseley. The first proponent of a modern form Periodic Table was Mendeleev who proposed what we would now regard as a short form. These advances would have been impossible without the wider determination of many atomic weights relative to hydrogen which was assigned the atomic weight of 1 and the recognition that some elements can exhibit more than one combining weight, an expression of what we would now understand is due to variable valency. He noted that groups of related elements are separated in weight order by seven other elements, and he even drafted a form of periodic table, though essentially with periods and groups at right angles to the modern usage. The recognition of weight relationships was extended further by Newlands who enunciated his Law of Octaves. In such triads the atomic weight of the middle element is roughly the mean of the atomic weights of the other two members. Döbereiner was an early systematiser who recognised the existence of triads, such as lithium + sodium + potassium, chlorine + bromine + iodine, and calcium + barium + strontium as early as 1817. They are not related to the real weights of atoms. These were then relative combining weights, and are essentially those still used today. It encouraged a more quantitative approach to inorganic chemical studies and the appearance of lists of so-called atomic weights. The general acceptance of atomic theory, often ascribed to the influence of Dalton who published his ideas in 1805, was the beginning of the modern scientific systemization of the inorganic chemistry. See back cover of this issue.Īttempts to systematize the chemistry of the elements in a logical fashion are more than 200 years old. Figure 1 Typical element entries taken from a long form of the Periodic Table.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
